|
Miller AC, Comellas AP, Hornick DB, Stoltz DA, Cavanaugh JE, Gerke AK, Welsh MJ, Zabner J, Polgreen PM. Identification of the cystic fibrosis gene: genetic analysis. Kerem B, Rommens JM, Buchanan JA, Markiewicz D, Cox TK, Chakravarti A, Buchwald M, Tsui LC 1989. Watch this video about Cystic Fibrosis Mechanism and Treatment (from HHMI BioInteractive). Like Kalydeco, Orkambi costs about $300,000 per year. This therapy combines ivacaftor with a second drug lumacaftor, which increases the trafficking of the ΔF508 protein to the cell surface. 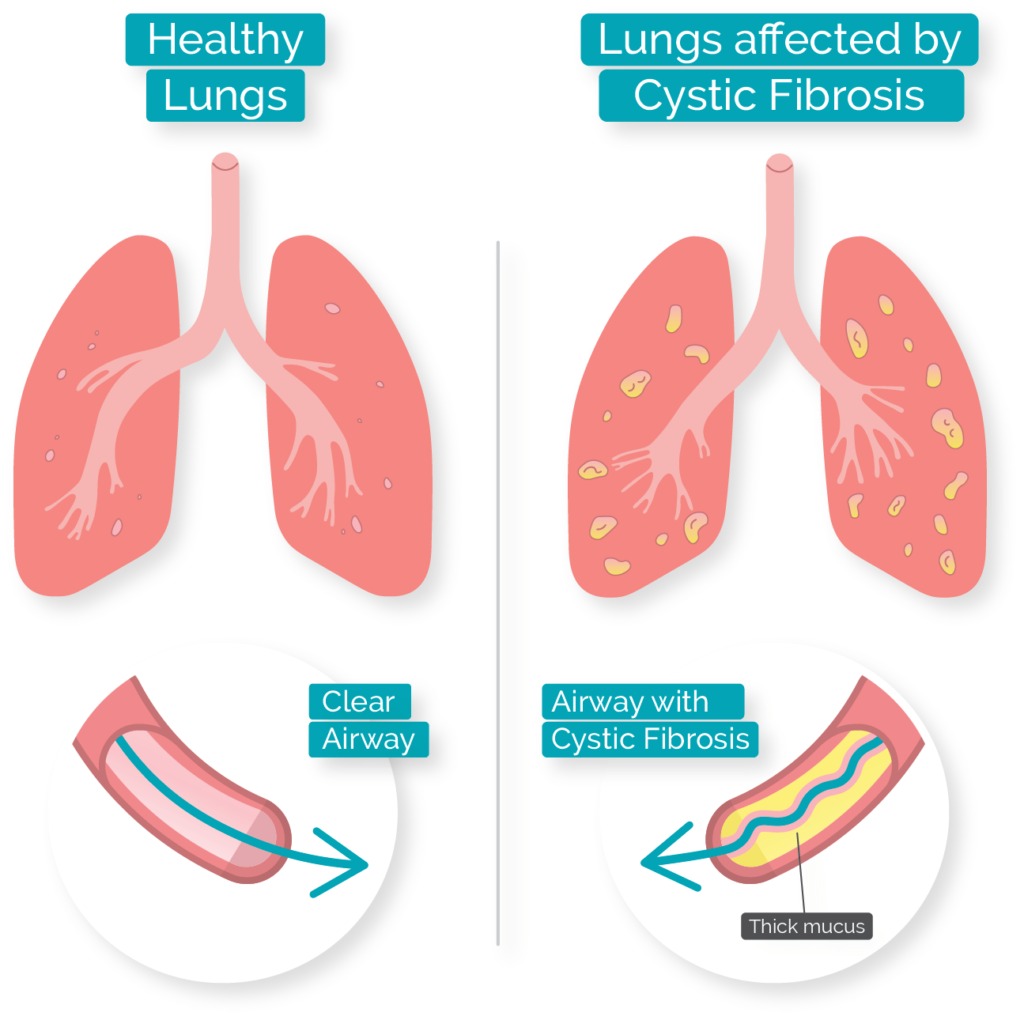
In 2015 an additional drug, Orkambi® was approved. Kalydeco® is priced at approximately $300,000 per patient per year, but costs may be covered by insurance. Kalydeco® compensates for the mutation by binding to CFTR and holding it in an open conformation. The G551D mutation affects the ability of ATP to bind to CFTR and open the channel it for transport. This mutation is found in less than 5% of CF patients. where glycine is substituted by aspartic acid at position 551 of the protein GLY551ASP). Kalydeco is effective on only some CFTR mutations, for example G551D (i.e. 1985 White et al., 1985) and cloned (Kerem et al. The drug, Kalydeco® (ivacaftor) made by Vertex Pharmaceuticals, was approved by the FDA and Health Canada in 2012, decades after the CFTR gene was first mapped to DNA markers (Tsui et al. However, recent mining of health data and genetic data from over 19,000 CFTR mutant allele heterozygotes suggests that heterozygotes exhibit an increased incidence of a variety of conditions including infertility, pancreatitis, diabetes, short stature, failure to thrive, constipation and scoliosis (Miller et al., 2020).ĬFTR is also notable because it is one of the well-characterized genetic diseases for which a drug has been developed that compensates for the effects of a specific mutation. The high frequency of the ΔF508 allele around the world has led to speculation that it may confer some selective advantage to heterozygotes, perhaps by reducing dehydration during cholera epidemics or by reducing susceptibility to certain pathogens that bind to epithelial membranes, although little data has been uncovered to support these hypotheses. The allele ΔF508 accounts for approximately 70% of CF cases in North America, with ~1/25 people of European descent being carriers. Mutation ΔF508 causes CFTR to be folded improperly in the endoplasmic reticulum (ER) and preventing CFTR from reaching the cell membrane. Worldwide, the most common CFTR allele among CF patients is called ΔF508 (delta-F508 or PHE508DEL), which is a deletion of three nucleotides that eliminates a phenylalanine (F) from position 508 of the 1480 amino acid wild-type protein. Any mutation that prevents CFTR from sufficiently transporting ions can lead to cystic fibrosis (CF). Over one thousand different mutant alleles of CFTR have been described. In some CFTR mutants, the channel does not open, which prevents the movement of ions and water and allows mucus to build up on the lung epithelium. /medriva/media/post_banners/content/uploads/2023/12/cystic-fibrosis-miracle-drugs-pricing-20231215140259.jpg)
\): Wild-type and mutant forms of CFTR in the cell membrane: In wild-type, the CFTR ion channel is gated when activated by ATP, the channel opens and allows ions to move across the membrane.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
